On December 14, 2020, the China National Intellectual Property Administration (CNIPA) released the Announcement of the CNIPA on Amending the Patent Examination Guidelines (No. 391) (国家知识产权局关于修改《专利审查指南》的公告(第391号)). This set of amendments focuses on pharmaceutical and biotech patent applications. The amended Guidelines go into effect on January 15, 2021.
A brief summary of the some of the amendments follow.
Supplementary Experimental Data
The Guidelines allow for the examination of supplementary experimental data after the filing date. “The technical effects proved by the supplementary experimental data should be obtained by those skilled in the art from the disclosure of the patent application.” CNIPA provides examples such as Example I:
Compound A is claimed. The description describes the preparation examples of compound A, the blood pressure lowering effect and the experimental method for measuring the blood pressure lowering activity, but the experimental result data is not recorded. In order to prove that the specification is fully disclosed, the applicant supplemented the blood pressure lowering effect data of Compound A. For those skilled in the art, according to the original application documents, the blood pressure-lowering effect of Compound A has been disclosed, and the technical effects to be proved by the supplementary experimental data can be obtained from the disclosure of the patent application documents. It should be noted that the supplementary experimental data should also be reviewed when examining for inventiveness.
Inventiveness of Chemical Compounds
The amendments rewrite Section 6.1, Chapter 10, Part Two of the “Patent Examination Guidelines” and adds examples.
6.1 Creativity of the compound
(1) To judge the inventiveness of a compound invention, it is necessary to determine the structural difference between the claimed compound and the compound closest to the prior art, and determine the technical problem actually solved by the invention based on the use and/or effect obtained by such structural modification. On this basis, it is judged whether the existing technology as a whole provides technical enlightenment for solving the technical problem through this structural transformation.
It should be noted that if a person skilled in the art can carry out this structural modification to solve the technical problem and obtain the claimed compound only through logical analysis, reasoning or limited experiment on the basis of the existing technology , it is believed that the existing technology has technical enlightenment.
(2) The use and/or effect brought about by the structural modification of the compound closest to the prior art by the invention can be to obtain a different use from the known compound, or it can be an improvement in a certain aspect of the effect of the known compound. When judging the inventiveness of a compound, if the change in use and/or the improvement of the effect is unexpected, it reflects that the claimed compound is non-obvious and its inventiveness should be recognized.
(3) It should be noted that when judging the inventive step of a compound invention, if the effect of the claimed technical solution is caused by a known inevitable trend, the technical solution is not inventive. For example, an insecticide AR in the prior art, wherein R is a C1-3 alkyl group, and it has been pointed out that the insecticidal effect increases as the number of C atoms of the alkyl group increases. If the insecticide applied for a certain application is A-C4H9, the insecticidal effect is significantly higher than that of the prior art. Since the prior art pointed out the inevitable trend of improving the insecticidal effect, the application is not inventive.
(4) Examples of inventiveness judgment
【example 1】
current technology:
(Ia)
Application:
(Ib)
The core structures of (Ib) and (Ia) are different, but both have the same purpose. Those skilled in the art generally believe that compounds with close structures have the same or similar uses, and close structures generally mean that the compounds have the same basic core part or basic ring. In the prior art, there is no technical enlightenment that the basic ring of (Ia) can be modified to obtain (Ib) and the use remains unchanged, so (Ib) is inventive.
【Example 2】
Existing technology: H2N-C6H4-SO2NHR1 (IIa)
Application: H2N-C6H4-SO2-NHCONHR1 (Ⅱb)
(IIb) is the insertion of -CONH- in the (IIa) NHR1 structural fragment. The two uses are completely different. (IIa) Sulfonamide is an antibiotic, (IIb) Sulfonylurea is an antidiabetic drug. Those skilled in the art have no motivation to transform R1 in antibiotics into CONHR1 to obtain antidiabetic drugs, so (IIb) is inventive.
【Example 3】
Existing technology: H2N-C6H4-SO2NHCONHR1 (Ⅲa)
Application: H3C-C6H4-SO2NHCONHR1 (Ⅲb)
(Ⅲa) Amino-sulfonylurea and (Ⅲb) methyl-sulfonylurea only differ in the structure of NH2 and CH3. Both are antidiabetic drugs with equivalent effects. (Ⅲb) is relative to (Ⅲa). The technical field provides another anti-diabetic drug. Since NH2 and CH3 are classical univalent isosteres, those skilled in the art have the motivation to carry out this isostere replacement in order to obtain the same or equivalent antidiabetic activity, so (IIIb) is not inventive.
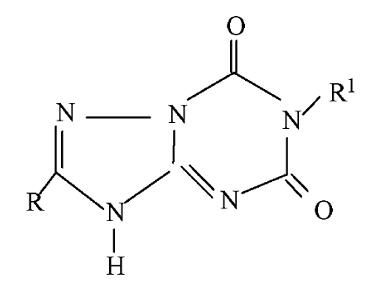
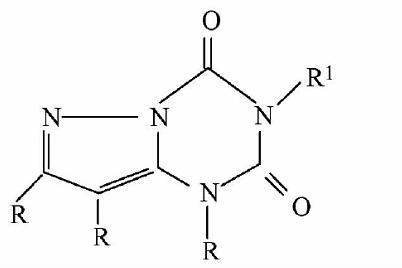
【Example 4】
current technology:
(IVa)
Application:
(IVb)
The difference between compound (IVb) and compound (IVa) is only that -O- replaces -NH- at the 6-position of the purine. Although -O- and -NH- are classical isosteres well-known in the art, the cancer cell growth inhibitory activity of (IVb) is about 40 times higher than that of (IVa), and (IVb) is expected to be higher than (IVa) The unobvious technical effect reflects that (IVb) is non-obvious, so (IVb) is inventive.
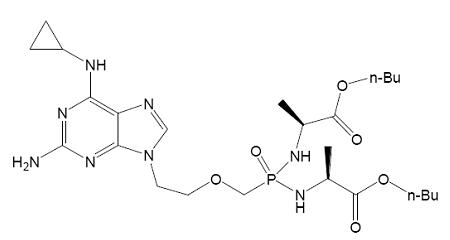
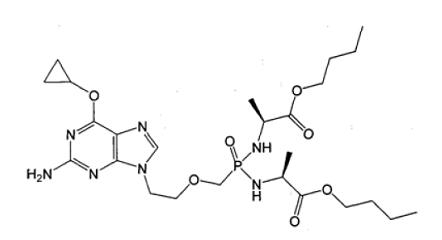
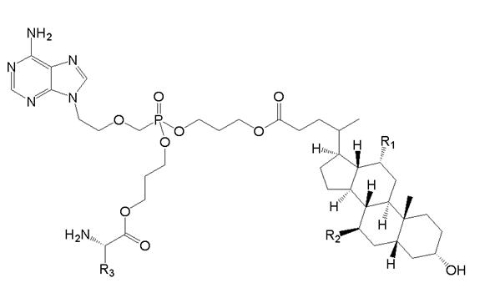
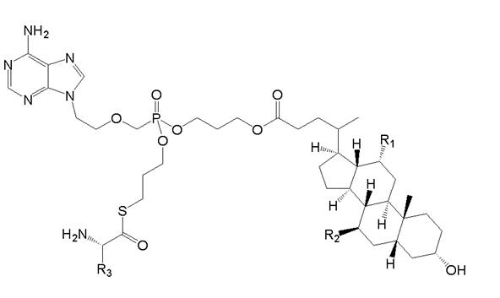
【Example 5】
current technology:
(Va)
Where R1=OH, R2=H and R3=CH2CH(CH3)2.
Application:
(Ⅴb)
Wherein R1 and R2 are selected from H or OH, R3 is selected from C1-6 alkyl, and includes specific compounds (Vb1) where R1=OH, R2=H and R3=CHCH3CH2CH3. And the anti-hepatitis B virus activity of (Vb1) is significantly better than (Va).
When the compound of the general formula (Vb) is claimed, the difference between (Vb) and (Va) is only the difference in the connecting atom between the phosphoryl alkyl group and the amino acid residue, (Vb) is -S-, and (Va) is -O-. Compared with (Va), the compound of formula (Vb) provides another anti-hepatitis B virus drug in the technical field. Since the properties of -S- and -O- are close, in order to obtain other drugs that also have anti-hepatitis B virus activity, those skilled in the art have the motivation to make this substitution and obtain the compound of the general formula (Vb), therefore (Vb) no inventiveness.
When the specific compound (Vb1) is claimed, the difference between (Vb1) and (Va) is not only the above-mentioned linking atom, but also the substituent at the R3 position. The anti-hepatitis B virus activity of (Vb1) is significantly better than that of (Va). There is no technical enlightenment to improve the anti-hepatitis B virus activity through the structural modification in the prior art, so (Vb1) is inventive.
Monoclonal Antibodies
The amendments rewrite Section 9.3.1.7, Chapter 10, Part Two:
Claims for monoclonal antibodies can be defined by structural features or by the hybridoma that produces it.
【E.g】
(1) A monoclonal antibody against antigen A, which comprises VHCDR1, VHCDR2 and VHCDR3 with amino acid sequences as shown in SEQ ID NO: 1-3, and VLCDR1, VLCDR2 and VLCDR3 with amino acid sequences as shown in SEQ ID NO: 4-6 .
(2) Monoclonal antibodies against antigen A are produced by hybridomas with the deposit number CGMCC NO:xxx.
Inventiveness in Biotechnology
Chapter 10, Section 9.4.2 of Part II is amended as follows:
Three new paragraphs are added under the title of Inventiveness, Chapter 10, Section 9.4.2 as follows:
To judge the inventiveness of an invention in the field of biotechnology, it is also necessary to judge whether the invention has outstanding substantive features and significant progress. In the judging process, it is necessary to determine the distinguishing features between the invention and the closest prior art based on the specific limitations, and then determine the technical problem actually solved by the invention based on the technical effect that the distinguishing feature can achieve in the invention. Determine whether the existing technology as a whole gives technical enlightenment, and based on this, determine whether the invention is obvious relative to the existing technology.
Inventions and creations in the field of biotechnology involve different levels of biology such as biological macromolecules, cells, and individual microorganisms. In the ways to characterize these biological entities, in addition to common ways such as structure and composition, there are also special ways such as the deposit number of biological materials. Inventive judgments need to consider the structural difference between the invention and the existing technology, the distance of the genetic relationship, and the predictability of technical effects.
The following shows some specific situations in the inventiveness judgments of different protection themes in this field.
The “Patent Examination Guidelines” Part Two, Chapter 10, Section 9.4.2.1, Item (1) is amended to:
(1) Gene
If a protein encoded by a structural gene has a different amino acid sequence and a different type or improved performance compared to a known protein, and the prior art does not provide the technical enlightenment that the sequence difference brings about the above performance change, then the gene invention encoding the protein is inventive.
If the amino acid sequence of a protein is known, the invention of the gene encoding the protein is not inventive. If a protein is known but its amino acid sequence is unknown, as long as those skilled in the art can easily determine its amino acid sequence when the application is filed, the gene invention encoding the protein is not inventive. However, in the above two cases, if the gene has a specific base sequence, and compared with other genes encoding the protein with a different base sequence, it has an effect that those skilled in the art would not expect, then the invention of genes is inventive.
If the structural gene claimed by an invention is a naturally available mutant structural gene of a known structural gene, and the claimed structural gene and the known structural gene are derived from the same species, they also have the same properties and functions , the invention is not inventive.
Add item (2) polypeptide or protein to Section 9.4.2.1, Chapter 10, Part Two of the “Patent Examination Guidelines”, as follows:
(2) Peptides or proteins
If the polypeptide or protein claimed by the invention is different from the known polypeptide or protein in amino acid sequence, and has different types or improved properties, and the prior art does not provide the technical enlightenment of the above-mentioned performance changes caused by the sequence difference, the invention of the polypeptide or protein is inventive.
Amend “(2) Recombinant Vector” in Section 9.4.2.1 of Chapter 10 of Part Two of the “Patent Examination Guidelines” to “(3) Recombinant Vector”, and insert a paragraph before the original content as follows:
If the invention aims at the structural modification of known vectors and/or inserted genes to improve the performance of the recombinant vector, and the prior art does not provide technical enlightenment for improving the performance of the above-mentioned structural modification, the invention of the recombinant vector is inventive.
Amend “(3) Transformation” in Section 9.4.2.1, Chapter 10, Part Two of the “Patent Examination Guidelines” to “(4) Transformation”, and insert a paragraph before the original content as follows:
If the invention aims at the structural modification of a known host and/or inserted gene to improve the performance of the transformant, and the prior art does not provide technical enlightenment for using the above-mentioned structural modification to improve the performance, the invention of the transformant is inventive.
Rewrite Section 9.4.2.1, Chapter 10, Part Two:
If the antigen is known, the monoclonal antibody of the antigen characterized by structural features is obviously different from the known monoclonal antibody in the key sequence that determines the function and use, and the prior art does not provide the monoclonal antibody that obtains the above sequence then if the monoclonal antibody can produce beneficial technical effects, the invention of the monoclonal antibody is inventive.
If the antigen is known, and it is clear that the antigen is immunogenic (for example, if the polyclonal antibody of the antigen is known or the antigen is a macromolecular polypeptide, it can be known that the antigen is obviously immunogenic), then the invention of monoclonal antibodies limited only with this antigen does not have an inventive step. However, if the invention is further limited by the hybridoma secreting the monoclonal antibody of the antigen, and thus produces unexpected effects, the invention of the monoclonal antibody is inventive.
The full text in comparison table format is available here: 专利审查指南 (Chinese only).



 />i
/>i