On Tuesday, Congressional leaders and the White House reached agreement on the Paycheck Protection Program and Healthcare Enhancement Act (H.R. 266), which was quickly taken up by the Senate in a pro forma session and passed by unanimous consent. The agreement is an intermediate relief package to supplement the “Phase 3” Coronavirus Aid, Relief, and Economic Security (CARES) Act enacted on March 27 (additional information available here). The $484 billion measure provides additional funding to the Small Business Administration (SBA) for the Paycheck Protection Program (PPP) and disaster loans and grants for small businesses; emergency relief for hospitals; and resources dedicated to coronavirus testing.
The bill is expected to be approved by the House of Representatives on Thursday and signed into law by the President shortly thereafter.
Missing from the latest package are infrastructure or other broad stimulus provisions that have been the primary subject of discussion for a potential “Phase 4” economy-wide stimulus bill. Funding for state and local governments and nutrition assistance supported by Democratic leadership were also not included in the agreement.
Summary of Paycheck Protection Program and Health Enhancement Act
Division A: Small Business Programs
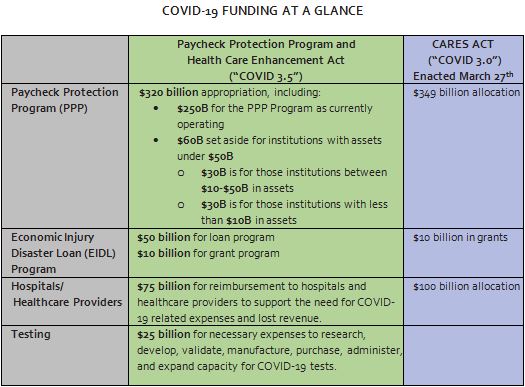
Paycheck Protection Program
The bill appropriates $320 billion for the PPP, bringing total program funding to $670 billion. The legislation sets aside $30 billion in loans made by Insured Depository Institutions and Credit Unions that have assets between $10 billion and $50 billion, and $30 billion for loans made by Community Financial Institutions, Small Insured Depository Institutions, and Credit Unions with assets less than $10 billion.
Other Small Business Administration Activities
The bill includes an additional $60 billion for emergency Economic Injury Disaster Loans (EIDLs), of which $50 billion is for loans and $10 billion is for grants administered by SBA. It also provides $2.1 billion to SBA to administer COVID-19 related programs.
As was the case in the CARES Act, the SBA is authorized to advance as much as $10,000 to EIDL recipients to pay sick leave to workers affected by COVID-19, retain employees, and make other covered payments. Advance funds do not have to be repaid. Additionally, this latest measure allows small agricultural businesses with 500 or fewer employees to obtain funds under both EIDL programs.
Division B: Additional Emergency Appropriations for Coronavirus Response
Hospitals
The bill provides $75 billion to reimburse hospitals and healthcare providers for COVID-19 related expenses and lost revenues.
Testing
The bill provides $25 billion to support research, development, validation, manufacturing, purchasing, administration, and expanded capacity for COVID-19 tests. Within this funding, $11 billion is provided to states, localities, territories, and tribes to bolster testing, scaling up laboratory testing, contact tracing, and employer testing. $2 billion is provided to states consistent with the Public Health Emergency Preparedness grant formula, ensuring every states receives funding, and $4.25 billion is provided to areas based on relative numbers of COVID-19 cases.
The bill also requires any state, locality, territory, or tribe receiving funding under the bill to submit to the Department of Health and Human Services (HHS) a detailed plan that sets goals for the remainder of 2020. These goals include:
- The number of tests required on a monthly basis;
- Monthly estimates of laboratory testing capacity; and
- A description of how federal resources will be used for testing, including as it relates to easing COVID-19 community mitigation policies.
HHS would also be required to submit a report to Congress on the status of a strategic COVID-19 testing plan to assist states, localities, territories, and tribes. This plan must include estimates of test production, guidelines for testing, a strategy to increase domestic testing capacity, and an outline of the availability of federal resources.
Finally, the measure would also set aside the following:
-
$1.8 billion for the National Institutes of Health (NIH) to develop testing and accelerate research on rapid testing, plus $500 million for the National Institute of Biomedical Imaging and Bioengineering and $306 million for the National Cancer Institute;
-
$1 billion for the Centers for Disease Control and Prevention (CDC) for activities such as surveillance, epidemiology, public health data surveillance, contact tracing, analytics infrastructure modernization, and lab capacity expansion;
-
$1 billion for the Biomedical Advanced Research and Development Authority for research, manufacturing, and purchasing tests;
-
Up to $1 billion to cover testing costs for the uninsured population;
-
$600 million for community health centers and federally qualified health centers, as well as $225 million for rural health clinics;
-
$750 million provided to tribes, tribal organizations, and urban Indian health organizations in coordination with Indian Health Service; and
-
$22 million for the Food and Drug Administration (FDA) to support activities associated with diagnostic, serological, antigen, and other tests, and related administrative activities.
Next Steps
The enactment of the Paycheck Protection Program and Healthcare Enhancement Act allows Congress to turn its attention to a “Phase 4” bill that could include the aforementioned provisions not included in this package, along with a host of other issues. House and Senate staff has been working behind the scenes to craft proposals and solicit input on a broader recovery and stimulus bill. However, it is unlikely that there will be any movement on additional legislation before Congress is scheduled to be back in session on May 4. The state of the economy over the next several weeks will also determine whether there is a need for additional intermediate relief to support businesses or taxpayers.



 />i
/>i