On February 24, 2020, the U.S. Environmental Protection Agency (EPA) hosted a conference call to review certain provisions of the final rule on fees for the administration of the Toxic Substances Control Act (TSCA), particularly those related to EPA-initiated risk evaluations. EPA published a Federal Register notice on January 27, 2020, identifying the preliminary lists of manufacturers (including importers) of the 20 high-priority chemical substances for risk evaluation for which fees will be charged. 85 Fed. Reg. 4661. During the comment period, currently scheduled to end on March 27, 2020, manufacturers (including importers) are required to self-identify as manufacturers of a high-priority substance irrespective of whether they are included on the preliminary lists identified by EPA. Where appropriate, entities may avoid or reduce fee obligations by making certain certifications consistent with the final rule on fees for the administration of TSCA. Manufacturers (including importers) identified on the final lists will be subject to applicable fees.
The total fee for an EPA-initiated risk evaluation is $1.35 million, and EPA will split this fee among self-identified payers. EPA’s 2018 final rule offers no exemptions for chemicals manufactured or imported as an impurity, byproduct, in an article, or in trace amounts. This is a dramatic shift from other TSCA provisions, in which certain reporting obligations are not required for impurities, byproducts, or articles. The TSCA fees rule contains provisions that offer options for potentially impacted companies to opt out of the fees obligations. If a company is, for example, improperly included on the preliminary list, it can provide a certification statement to EPA that it has not manufactured or imported the subject chemical -- in any amount -- over the past five years. If a company manufactured or imported in the five years prior to the publication of a preliminary list of manufacturers, but ceased manufacture or import -- in any amount -- before the subject chemical was identified as a priority candidate, and agrees not to manufacture or import again for five years, the company will not be obligated to pay the fees. Unfortunately, if a company manufactured or imported any of the 20 high-priority chemicals after March 20, 2019, it cannot avoid the fee obligation.
Publication of the preliminary lists has inspired considerable confusion. During the conference call, Ryan Schmit, EPA, stated that EPA is strongly considering extending the March 27, 2020, deadline. According to Schmit, EPA is looking into options to help address or lessen concerns regarding the lack of exemptions. At this time, EPA is not in a position to specify additional guidance on issues such as identifying importers, but if EPA has additional guidance, it would be announced before EPA issues the final list. EPA intends to publish a final list of payers in approximately June 2020. Manufacturers and importers on the final list will be subject to the EPA-initiated risk evaluation fee. Schmit stated that EPA does not see another opportunity for comment after publishing the final list.
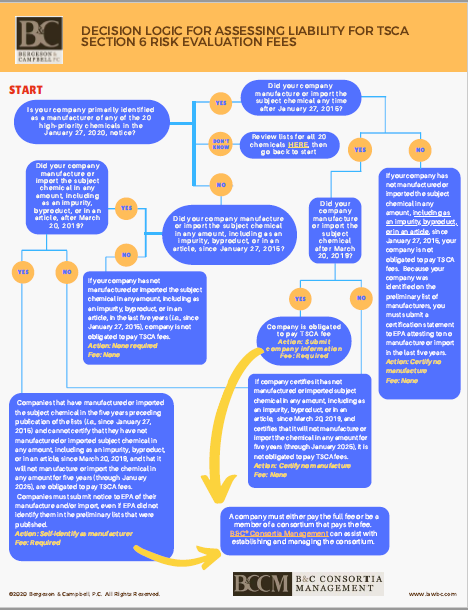
Bergeson & Campbell, P.C. (B&C®) has prepared the following decision tree for manufacturers and importers to use to assess their liability for the EPA-initiated risk evaluation fee; additional resources are available below the graphic.
Resources
Information about TSCA fees is available on EPA’s website. EPA has posted a transcript of the February 24, 2020, conference call, as well as a transcript of a December 19, 2019, conference call on TSCA fees associated with EPA-initiated risk evaluations.
The preliminary lists of manufacturers (including importers) of the 20 high-priority chemical substances for risk evaluation are available in Docket EPA-HQ-OPPT-2019-0677 and on EPA’s website. EPA states that it developed each preliminary list “using the most up-to-date information available, including information submitted to the Agency (e.g., information submitted under TSCA section 8(a) (including the Chemical Data Reporting (CDR) Rule) and section 8(b), and to the Toxics Release Inventory (TRI)).” To include the two most recent CDR reporting cycle data (collected every four years) and to account for annual or other typical fluctuations in manufacturing (including import), EPA states that it used six years of data submitted or available to it under CDR and TRI to create the preliminary lists (2012-2018).
The Notice of TSCA Fee Action Participation form is available in the docket for the final rule. EPA has stated that this form is for reference only. It does, however, provide context as to the response options anticipated under this EPA action.
More information on the 20 substances designated as high-priority substances is available in our December 20, 2019, memorandum, “Final List of High-Priority Chemicals Will Be Next to Undergo Risk Evaluation under TSCA.”
More information on the final TSCA fees rule is available in our September 28, 2018, memorandum, “EPA Issues Final TSCA Fees Rule.”


 />i
/>i