On 3 August 2020, the Centers for Medicare & Medicaid Services (CMS) released its Physician Fee Schedule (PFS) proposed rule (Proposed Rule) for Calendar Year (CY) 2021.1 The agency will accept comments on the Proposed Rule until 5:00 p.m. on 5 October 2020. In this alert, we highlight certain aspects of the Proposed Rule, including proposed changes to evaluation and management (E/M) services, care management services and remote physiologic monitoring (RPM) services, and supervision of diagnostic tests.
OFFICE/OUTPATIENT VISIT E/M SERVICES
CY 2021 will begin with CMS’s implementation of sweeping changes to the long-standing rules for E/M code selection that were outlined in last year’s PFS Final Rule.2
Since the inception of the E/M portion of the Current Procedural Terminology (CPT) code set in 1992, most E/M visit codes were based on the level of complexity, described broadly by the key components of history, exam, and medical decision-making (MDM), with contributory components for counseling, coordination of care, and nature of the presenting problem. While time was part of the CPT descriptor for most E/M codes, time was only a factor in determining the level of service when counseling and coordination of care dominated (>50 percent) of the face-to-face time spent between the provider and the patient.
In the CY 2019 PFS Final Rule, CMS finalized a number of changes specific to the Office/Outpatient E/M codes as part of its effort to reduce administrative burden, improve payment accuracy, and update this part of the code set. However, in early 2019, the American Medical Association (AMA)/CPT established the Joint AMA CPT Workgroup on E/M to develop an alternative solution. The approach developed by the Workgroup was presented to CMS after its approval by the CPT Editorial Panel in February 2019. As noted above, CY 2020 PFS Final Rule finalized CMS’s plan to adopt the AMA’s MDM guidelines and to use the AMA’s definitions of time or MDM to select office/outpatient codes effective 1 January 2021.3
-
99201 is being deleted, effective 1 January 2021.4
-
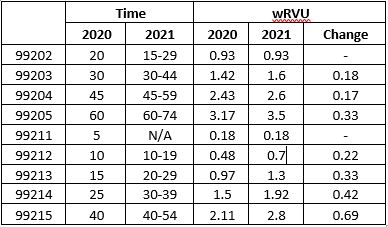
New patient office/outpatient visit services will be reported with CPT codes 99202-99205 o Established patient office/outpatient visit services will be reported with CPT codes 99211-99215, with 99211 being used to include E/M visits performed by clinical staff for established patients. For Medicare patients, 99211 can only be reported when the “incident to” requirements are also met.
-
-
The revised Office/Outpatient code definitions rely on total time or MDM for code selection. While history and exam will not be used to as part of the selection criteria for the level of service for these revised codes, CMS indicated a medically appropriate history and exam should still be documented when performed.
-
Time is defined in ranges for each code (see table below).
-
Time can be the basis for 99202-99215 code selection, even if counseling/coordination of care is not more than 50 percent of the face to face time.
-
-
CMS has adopted the AMA’s new MDM table that was defined specific to the new 99202-99215 code definitions (the CY 2021 Proposed Rule references the AMA website for more information on the new MDM table).5
Since the new time definitions for the revised Office/Outpatient visit codes include non-face-to-face time that occurs on the same calendar day (before and after the encounter), CMS will discontinue the use 99358-99359 (Prolonged Services without direct patient contact) codes for Office/Outpatient visits. There will be a new CPT prolonged services add-on code 99XXX that can be added to the E/M code once the time for the level 5 service code has been exceeded by at least 15 minutes.
CMS has proposed to create a Healthcare Common Procedure Coding System (HCPCS) add-on code GPC1X to address interactive complexity during an office/outpatient encounter.
CMS also proposes to revalue other services that include office/outpatient E/M equivalents in their relative value unit calculations. Some examples are end-stage renal disease (ESRD)-related visit services, transitional care management (TCM), emergency department visit codes, maternity services, several therapy evaluation codes and psychotherapy codes. A complete listing of codes and their proposed revaluations can be found on pages 50124-50137.
CARE MANAGEMENT SERVICES AND RPM SERVICES
Consistent with CMS policies to improve payment for care management, the Proposed Rule seeks to improve payment for such services by refining codes for RPM and TCM.6
RPM allows for the creation of a management treatment plan related to a chronic or, as clarified by CMS in the Proposed Rule, acute7 health illness or condition through analysis of a patient’s physiologic data. In the Proposed Rule, CMS asks for comments in regard to whether the current RPM coding accurately describes the full universe of clinical scenarios in which RPM services could be utilized.8 The Proposed Rule would also allow patient consent to be obtained at the time that RPM services are provided.9 CMS is proposing to allow auxiliary personnel (e.g., including individuals who are leased or contracted employees) to provide services described by CPT codes 99453 and 99454 under the general supervision of the billing physician or practitioner.10
In regard to TCM, the Proposed Rule proposes to remove 14 additional actively priced HCPCS codes from the list of remaining codes that cannot be billed concurrently with TCM and proposes to allow an add-on code, G2058 (base code 99490), to also be billed within the same calendar month with TCM when medically necessary. The Proposed Rule also identifies that the full range of ESRD codes (90951-90969) may be billed concurrently with TCM services when reasonable and necessary.11
SUPERVISION OF DIAGNOSTIC TESTS AND SCOPE OF PRACTICE PROPOSALS
During the Public Health Emergency (PHE), CMS added flexibility in 42 C.F.R. § 410.32(b) through its interim final rule to allow certain nonphysician practitioners (NPPs) to supervise diagnostic tests.12 Nurse practitioners, clinical nurse specialists, physician assistants, and certified nurse-midwives were among the NPPs included.
In the CY 2021 Proposed Rule, CMS proposes to make its interim policy permanent, to the extent the NPPs are authorized to supervise diagnostic tests under their state scope of practice and applicable state law.13 CMS notes that while the agency expects proposed scope of practice policies to increase administrative and clinical flexibility, it cannot determine the impact on related business plans or the demand for various types of physicians.14
PAYMENT FOR PRIMARY CARE MANAGEMENT SERVICES IN RHCS AND FQHCS
A separate payment for primary care management (PCM) services was added through CMS’s CY 2020 PFS Final Rule with comment.15 PCM services could be billed using HCPCS codes G2064 or G2065 beginning on 1 January 2020. However, for CY 2020, PCM services were not separately payable to rural health clinics (RHCs) or federally qualified health centers (FQHCs), as they did not meet the criteria for an RHC or FQHC billable visit.
CMS is now proposing to add HCPCS codes G2064 and G2065 to the list of services billed with G0511 as a comprehensive care management service for RHCs and FQHCs starting on 1 January 2021.16 CMS is proposing to revalue G0511 to calculate a new average for the national non-facility PFS payment rate, to be updated annually based on the PFS amounts for these codes, respectively. Services using G0511 will continue to be paid separately from the all-inclusive rate for an RHC or FQHC claims and, if finalized, will be paid at the new rate for dates of service on or after 1 January 2021.17
CONCLUSION
In addition to proposing policies that will impact payment rates and services furnished under Medicare, the CY 2021 PFS Proposed Rule indicates that regulatory changes could be on the horizon, such as changes to professional scope of practice for certain NPPs. K&L Gates’ health care practice regularly advises clients in the area of Medicare reimbursement and continues to monitor developments surrounding CMS’s rulemaking, guidance, and policies. Contact the authors of this article or your K&L Gates lawyer for assistance with submitting comments to CMS on the Proposed Rule or to learn more about the potential impact of the changes outlined in this alert.
Footnotes
1 Medicare Program; CY 2021 Payment Policies Under the Physician Fee Schedule and Other Changes to Part B Payment Policies; Medicare Shared Savings Program Requirements; Medicaid Promoting Interoperability Program Requirements for Eligible Professionals; Quality Payment Program; Coverage of Opioid Use Disorder Services Furnished by Opioid Treatment Programs; Medicare Enrollment of Opioid Treatment Programs; Electronic Prescribing for Controlled Substances for a Covered Part D Drug Under a Prescription Drug Plan or an MA–PD Plan; Payment for Office/Outpatient Evaluation and Management Services; Hospital IQR Program; Establish New Code Categories; and Medicare Diabetes Prevention Program (MDPP) Expanded Model Emergency Policy, 85 Fed. Reg. 50074 (Aug. 17, 2020). [hereinafter CY 2021 PFS].
2 Id. at 50121-50124.
3 Changes in definition and documentation requirements only apply to Evaluation/Management codes for Office/Outpatient visits. All other subcategories of E/M codes will continue to follow the historical definitions and documentation requirements that have been in place since the 1990s.
4 CY 2021 PFS, 85 Fed. Reg. 50074, 50122.
5 Id. See also Table 2 “Level of Medical Decision Making (MDM).” American Medical Association, CPT® Evaluation and Management (E/M) Office or Other Outpatient (99202-99215) and Prolonged Services (99354, 99355, 99356, 99XXX) Code and Guideline Changes (last accessed on August 21, 2020) pgs. 7-8.
6 See Table 13, “Summary of Care Management Codes.” CY 2021 PFS, 85 Fed. Reg. 50117.
7 CY 2021 PFS, 85 Fed. Reg. 50074, 50117.
8 Id.
9 Id.
10 Id.
11 See Table 14, “15 Additional Codes that Could Be Billed with TCM.” CY 2021 PFS, 85 Fed. Reg. 50120.
12 Medicare and Medicaid Programs, Basic Health Program, and Exchanges; Additional Policy and Regulatory Revisions in Response to the COVID–19 Public Health Emergency and Delay of Certain Reporting Requirements for the Skilled Nursing Facility Quality Reporting Program, 85 Fed. Reg. 27550, 27555 (May 8, 2020).
13 CY 2021 PFS, 85 Fed. Reg. 50074, 50377.
14 Id.
15 See 84 Fed. Reg. 62568, 62692 (Nov. 15, 2019).
16 CY 2021 PFS, 85 Fed. Reg. 50074, 50214.
17 Id.



 />i
/>i